
In addition, alternative pre-mRNA splicing results in the production of isoforms (DNA ligases IIIα and IIIβ with different carboxy-terminal sequences. The DNA ligase III gene uses a similar mechanism to produce nuclear and mitochondrial proteins. The nuclear form of the protein, which lacks the MTS, is translated from an internal in-frame AUG. This pre-protein is localized to the mitochondria, whereupon the MTS is cleaved by a mitochondrial peptidase. Translation from the first AUG gives rise to a pre-protein with an amino-terminal mitochondrial targeting sequence (MTS). In budding yeast, the use of different start codons results in the translation of distinct nuclear and mitochondrial iso-forms of the DNA ligase I protein Cdc9. The PBM is found at the amino terminus of nuclear DNA ligase I enzymes from yeast and vertebrates, as well as in a number of other DNA replication and repair factors, such as the large subunit of the 'clamp loader' replication factor C (RF-C), which loads PCNA onto DNA, and the nuclease FEN1. PCNA (proliferating cell nuclear antigen) is best known as a DNA polymerase processivity factor, but there is increasing evidence that it plays an important role in coordinating protein-protein interactions on DNA. In addition to the CD and NCD domains, nuclear DNA ligase I proteins from different species also have an amino-terminal domain of variable length and low sequence conservation that includes a nuclear localization sequence (NLS) and, at the extreme amino terminus, a conserved PCNA-binding motif (PBM) of the type first identified in the mammalian DNA replication inhibitor p21 Cip1. These studies have identified a vital role for this enzyme in nuclear DNA repair. Like DNA ligase I, ligase IV is also likely to be conserved in all eukaryotes: to date, orthologs of DNA ligase IV have been identified and characterized in yeast, higher plants and vertebrates. This latter enzyme, which to date has been identified only in vertebrates, is also present in the nucleus, where it functions in DNA repair and perhaps also in meiotic recombination. In budding yeast a form of DNA ligase I also functions in mitochondrial DNA replication and repair, a role that in higher eukaryotes is taken by DNA ligase III. DNA ligase I is probably conserved in all eukaryotes: orthologs have been identified and characterized in organisms as diverse as yeast and mammals, and have been shown to play important roles in nuclear DNA replication, repair and recombination. Vertebrate cells encode three well-characterized DNA ligases - DNA ligases I, III and IV - that appear to be descended from a common ancestral nucleotidyltransferase enzyme. Recent structural studies have shed light on the catalytic function of DNA ligases, as well as illuminating protein-protein interactions involving DNA ligases IIIα and IV. DNA ligase IV forms a tight complex with Xrcc4 through an interaction motif located between a pair of carboxy-terminal BRCT domains in the ligase. DNA ligase IV is required for DNA non-homologous end joining pathways, including recombination of the V(D)J immunoglobulin gene segments in cells of the mammalian immune system. 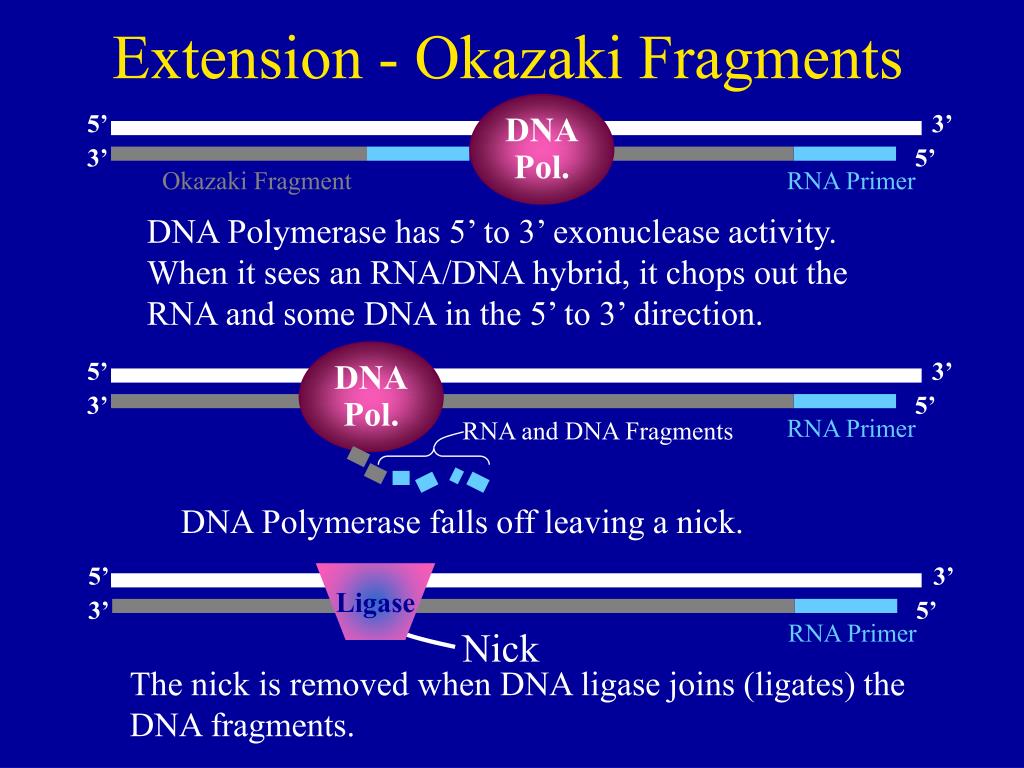
Two distinct isoforms of this enzyme, differing in their carboxy-terminal sequences, are produced by alternative splicing: DNA ligase IIIα has a carboxy-terminal BRCT domain that interacts with the mammalian DNA-repair factor XrccI, but both α and β isoforms have an amino-terminal zinc-finger motif that appears to play a role in the recognition of DNA secondary structures that resemble intermediates in DNA metabolism. DNA ligase III, which is unique to vertebrates, functions both in the nucleus and in mitochondria. DNA ligase I is required for the ligation of Okazaki fragments during lagging-strand DNA synthesis, as well as for several DNA-repair pathways these functions are mediated, at least in part, by interactions between DNA ligase I and the sliding-clamp protein PCNA. Enzymes of each class comprise catalytic and non-catalytic domains together with additional domains of varying function. Three related classes of ATP-dependent DNA ligase are readily apparent in eukaryotic cells. By catalyzing the joining of breaks in the phosphodiester backbone of duplex DNA, DNA ligases play a vital role in the diverse processes of DNA replication, recombination and repair.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
